I. Material Composition of Lithium-ion Batteries
Lithium-ion (Li-ion) batteries are currently the most prevalent energy storage solution for New Energy Vehicles (NEVs). A standard cell consists primarily of a cathode, an anode, a separator, and electrolyte.
1.1 Cathode Materials
The cathode is the most critical component of a Li-ion battery, accounting for over 40% of the total cell cost. Its performance directly dictates the battery’s key specifications. The development of high-energy-density cathodes remains a primary bottleneck in achieving higher energy, longer cycle life, and lower costs.
- Lithium Cobalt Oxide (LiCoO2 / LCO): One of the most established materials on the market. It features a 2D layered structure ideal for ion intercalation. While its theoretical capacity is 274 mAh/g, structural stability limits actual capacity to approximately 140 mAh/g at an average operating voltage of 3.7V. It is typically produced via high-temperature solid-state synthesis—a process favored for mass production due to its simplicity and the material’s excellent electrochemical stability.
- Lithium Nickel Oxide (LiNiO2 / LNO): Shares a similar cubic rock-salt structure with LCO but is more cost-effective. It offers a higher practical capacity (140–180 mAh/g) and an operating range of 2.5V–4.2V. Despite its high discharge capacity and thermal stability, it is difficult to synthesize and requires strict oxygen-rich conditions, leading to limited commercial use.
- Lithium Manganese Oxide (LMO): Typically found in two structures: orthorhombic (LiMnO2) and spinel (LiMn2O4). Spinel LMO is more common, offering a 4.0V high-voltage platform and superior safety/overcharge resistance, with a capacity (148 mAh/g) comparable to LCO.
- Lithium Iron Phosphate (LiFePO4 / LFP): Features an olivine structure. LFP is renowned for its exceptional cycle life and thermal stability. While it approaches its theoretical capacity (170 mAh/g) at room temperature, it suffers from high polarization at high C-rates and lower conductivity.
- Nickel-Cobalt-Manganese (NCM/Ternary): These materials leverage a synergistic effect: Nickel increases capacity, Cobalt stabilizes the layered structure, and Manganese enhances safety while reducing costs. Ternary cathodes offer superior overall performance but require precise stoichiometric balancing.
1.2 Anode Materials
The anode acts as the host for lithium ions and electrons during charging, facilitating energy storage and release. It typically accounts for 5%–15% of the total cell cost.
- Graphite Anodes: The industry standard. Graphite has a theoretical capacity of 372 mAh/g. During the initial charge, a Solid Electrolyte Interphase (SEI) layer forms on the surface due to electrolyte decomposition. While graphite is stable, it experiences about 10% volume expansion during intercalation, and its low potential can lead to slower charging rates.
- Silicon-based Anodes: With a massive theoretical capacity of 4,200 mAh/g, silicon is the “next frontier.” However, unlike graphite’s intercalation, silicon undergoes an alloying reaction that causes significant volume expansion (up to 300%). This leads to mechanical failure, loss of electrical contact, and continuous SEI reconstruction, which rapidly degrades cycle life.
- Lithium Titanate (LTO): While offering high safety and power, LTO and metallic lithium anodes still face challenges regarding energy density and dendritic growth.
1.3 Electrolyte
The electrolyte serves as the medium for ion transport between the cathode and anode. It is essential for achieving high voltage and high specific energy, representing about 15% of the cell cost.
- Types: Electrolytes are categorized into liquid (inorganic/organic), solid (pure polymer/gel), and molten salts.
- Lithium Salts: Lithium Hexafluorophosphate (LiPF6) is the most common salt due to its high conductivity, solubility, and relative stability.
1.3.1 Additives
Additives are “secret sauce” formulas used to enhance specific performance metrics:
- Film-forming additives: Promote a stable SEI layer to prevent electrolyte breakdown.
- Conductivity enhancers: Improve ion mobility and salt dissociation.
- Flame retardants: Increase thermal stability and prevent ignition.
- Overcharge protection: Use redox shuttles to prevent damage when the voltage exceeds safety limits.
- Low-temperature additives: Ensure the battery remains functional and efficient in cold climates.
1.4 Separator
The separator is a microporous membrane that prevents physical contact (electrical shorting) between the electrodes while allowing lithium ions to pass through. It is a safety-critical component. Most commercial separators are made of polypropylene (PP) or polyethylene (PE) microporous films due to their excellent chemical stability and mechanical strength.
1.5 Current Collectors
Current collectors—typically Copper foil for the anode and Aluminum foil for the cathode—facilitate electron flow to the external circuit.
- Copper is preferred for the negative electrode due to its high conductivity, flexibility, and stability at low potentials.
- Aluminum is used for the positive electrode and as a barrier in pouch cell packaging. Both must ensure low internal resistance and strong adhesion to active materials.
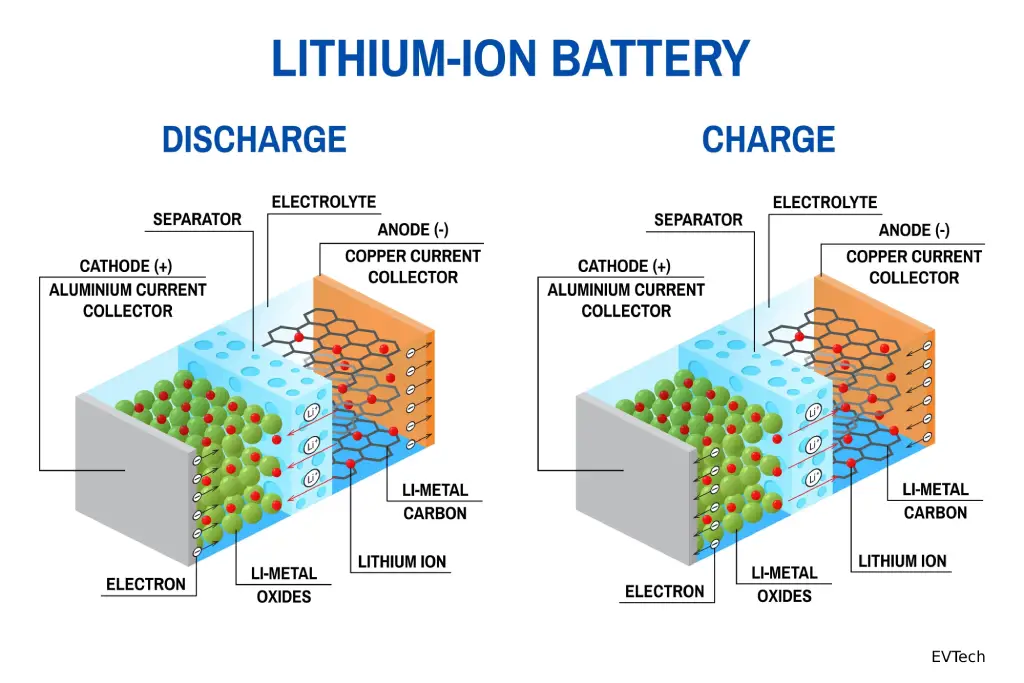
II. The Charge and Discharge Process
A Li-ion battery operates on the principle of a concentration gradient, where ions shuttle back and forth between the electrodes.
2.1 Charging
During charging, lithium ions are “de-intercalated” from the cathode, migrate through the electrolyte and separator, and are embedded into the anode. The anode becomes “lithium-rich” while electrons flow through the external circuit to maintain charge neutrality.
2.2 Discharging
The process reverses: ions move from the anode back to the cathode. This migration causes electrons to flow from the anode through the copper current collector to power the external load.
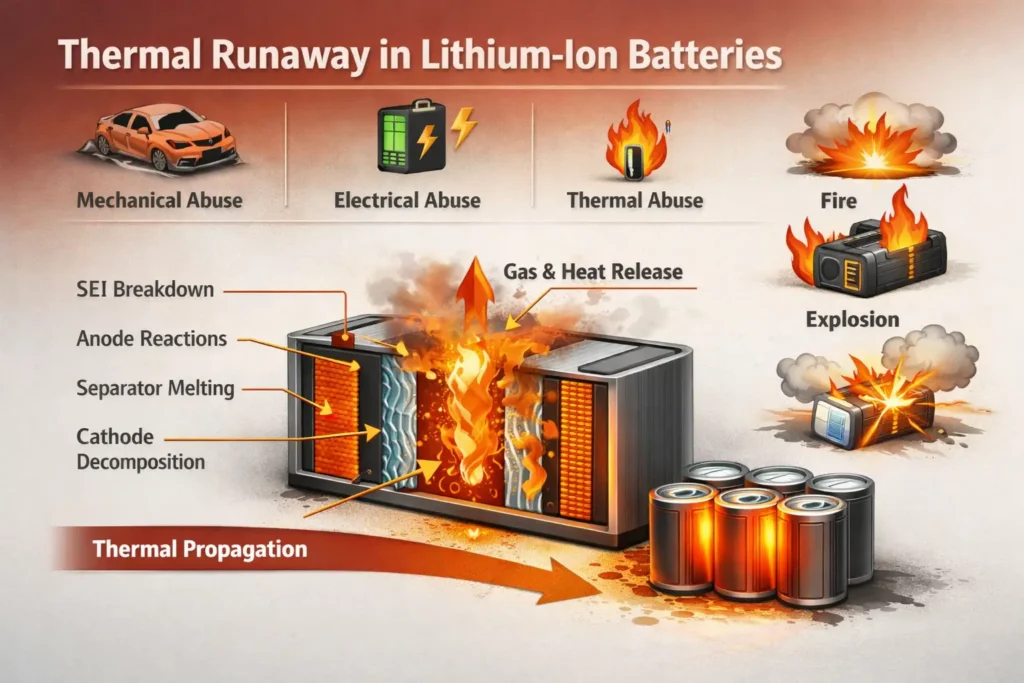
2.3 Irreversible Loss and Safety
In an ideal scenario, this cycle is perfectly reversible. In reality, batteries suffer from irreversible consumption such as electrolyte decomposition, active material dissolution, and lithium plating. Over time, these losses reduce capacity and can lead to safety risks. Extreme conditions—such as thermal shock, overcharging, or short circuits—can trigger thermal runaway, leading to fires or explosions.
Because of these risks, material selection is paramount. Furthermore, real-time monitoring via a Battery Management System (BMS) is essential to ensure the longevity and safety of the pack—a topic we will explore in future installments.